PROPPR

PROPPR: Transfusion of Plasma, Platelets, and Red Blood Cells in a 1:1:1 vs a 1:1:2 Ratio and Mortality in Patients With Severe Trauma. The PROPPR Randomized Clinical Trial
PROPPR Study Group*, JAMA. 2015;313(5):471-482
Clinical Question
-
In patients with severe trauma and predicted to require massive transfusion, does the use of a transfusion protocol using a 1:1:1 ratio of plasma to platelets to red blood cells (RBCs) compared to 1:1:2 improve mortality?
Design
- Randomised-controlled trial
- Treating clinicians non-blinded
- Allocation concealment maintained through use of sealed containers of blood products
- Blinding of assessors of primary outcome
- Sample size calculation
- Sample size increased from 580 to 680 patients according to “trial’s adaptive design” due to lower actual mortality than predicted
- With sample size of 680 patients
- 95% power to detect 10% absolute difference in mortality at 24 hours
- 92% power to detect 12% absolute difference in mortality at 30 days
Setting
- 12 North American Level 1 trauma center sites
- 3rd August 2012 – 2nd December 2013
Population
- Inclusion:
- Highest trauma level activation
- Estimated age 15 years or older or greater than/equal to weight of 50 kg if age unknown
- Received directly from scene of injury
- Received at least 1 U of blood product in pre-hospital setting or within 1 hour of arrival to hospital
- Predicted to receive a massive transfusion by Assessment of Blood Consumption Score ≥ 2 or by the attending trauma surgeon’s judgment
- Exclusion:
- Received a life saving intervention from another hospital
- Non-survivable injuries
- Prisoners directly admitted from jail
- Required emergency thoracotomy prior to randomisation
- Obvious pregnancy
- Severely burned
- Had at least 5 minutes of CPR with chest compressions before admission
- Known “Do Not Resuscitate” orders prior to randomization
- Enrolled in a concurrent ongoing interventional, randomized clinical trial
- Patients who wear “opt-out” bracelet
- >3 U RBCs given before randomization
- PROPPR products not given within 2 hour period
- Patient improved, did not require further transfusion
- 11185 screened, 680 patients randomised (338 to intervention, 342 to control)
- Groups eventually matched including Injury Severity Scores and Revised Trauma Scores (median ISS score 26)
Intervention
- Blood product ratios of 1:1:1 (plasma:plts:RBCs)
- Packs contained 6 U plasma, 1 dose plts (pool of 6 U) and 6 U RBCs
Control
- Blood product ratios of 1:1:2 (plasma:plts:RBCs)
- Alternating packs containing 3 U plasma, 0 dose plts and 6 U RBCs with 3 U plasma, 1 dose plts and 6 U RBCs
In both intervention and control group
- Blood products administered in a pre-specified order to maintain assigned ratios
- Transfusion stopped when clinically indicated
Outcome
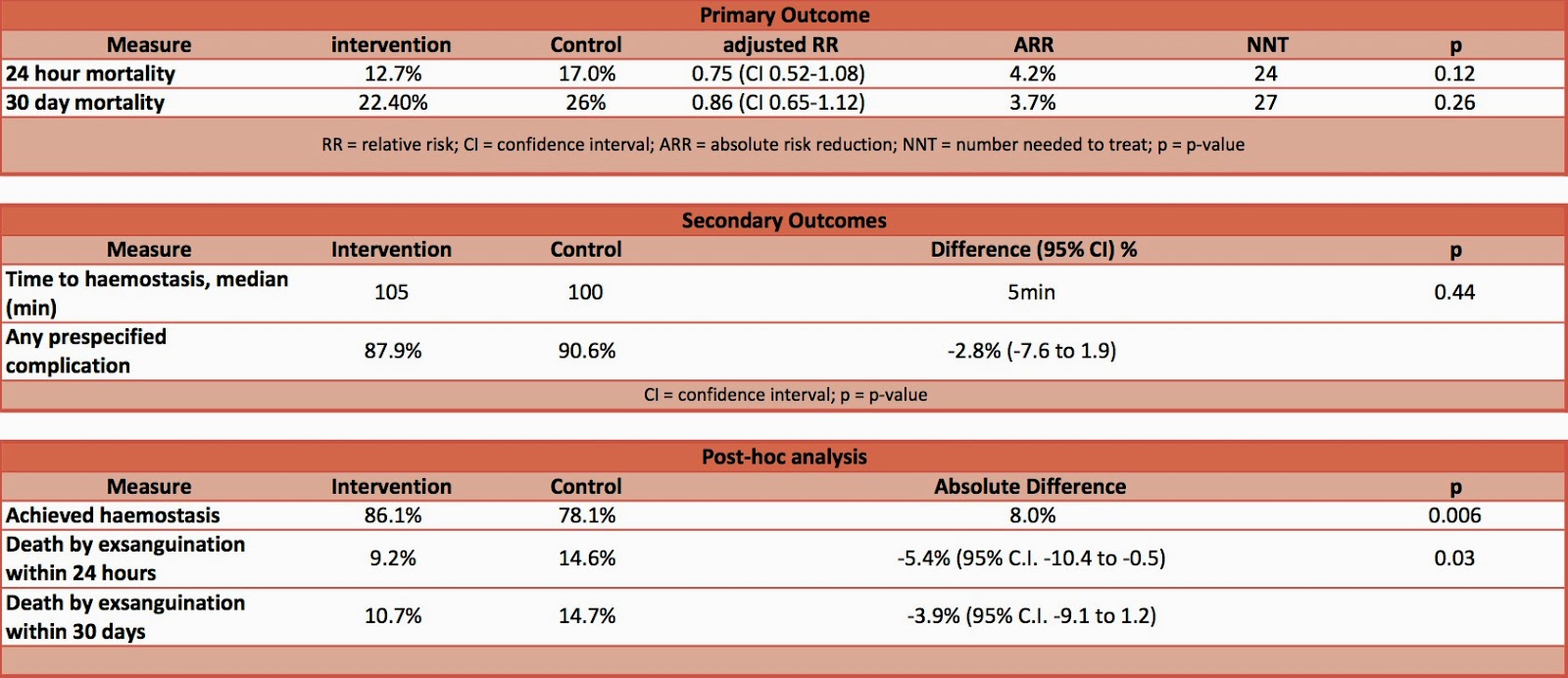
- Primary outcome:
- 24 hour mortality – no significant difference
-
30 day mortality – no significant difference
- Secondary outcome:
- No significant difference in:
- Time to haemostasis
- Any of 23 pre-defined complications
- Hospital-, ventilator, and ICU-free days
- Incidence of surgical procedures
- Functional status at discharge
- Blood product volumes transfused
- Until haemostasis achieved – no significant difference
- After haemostasis till 24 hours post-admission – significantly higher in intervention group
- Post-Hoc Analysis
- Death by Exsanguination in 1st 24 hours– significantly decreased in intervention group
- Achieved haemostasis – significantly greater in intervention group
- No significant difference in:
Authors’ Conclusions
- Among patients with severe trauma and major bleeding, early administration of plasma, platelets, and red blood cells in a 1:1:1 ratio compared with a 1:1:2 ratio did not result in significant differences in mortality at 24 hours or at 30 days.
Strengths
- Randomised, multi-centre, pragmatic
- Targeted most severely injured trauma patients predicted to be at highest risk of haemorrhage
- Pre-specified transfusion order to maintain target ratios
- Sample size increased to maintain statistical power
- Minimal loss during follow up
- Allocation concealment contained
Weaknesses
- Unblinded
- 30 day maximum follow up time
- Powered to detect an absolute difference of 10% in mortality – unable to detect smaller effect
- 2968 patients would have been required to detect the observed difference of 4.2% (24 hour mortality) with 90% power
The Bottom Line
- Among patients with severe trauma and major bleeding there was no significant difference in 24 hour or 30 day mortality in patients who received a blood product transfusion at a ratio of 1:1:1 compared to a ratio of 1:1:2. However the study was significantly underpowered for the observed difference in mortality found.
- A ratio of 1:1:1 resulted in reduced mortality from exsanguination within the 1st 24 hours, with similar complication rates to a lower ratio protocol. Therefore, I will continue to aim for a 1:1:1 ratio in this population.
External Links
- [Article – abstract & supplementary data] ] Transfusion of Plasma, Platelets, and Red Blood Cells in a 1:1:1 vs a 1:1:2 Ratio and Mortality in Patients With Severe Trauma
- [Podcast – FOAMed] EMCrit Podcast 13 – Trauma Resus II: Massive Transfusion
- [RebelEM – FOAMed] REBELEM PROPPR
- [EMNerd – FOAMed] EM Nerd
- [EM Literature of Note – FOAMed] EMLitofNote
- [MDCalc] Assessment-of-blood-consumption-for-massive-transfusion
- [St Emlyn’s] Getting the Balance Right: The PROPPR Trial
- [SGEM] SGEM #109 One Platelet, One Plasma and One RBC – PROPPR Trial
Metadata
Summary author: @avkwong
Summary date: 7th Feb 2015
Peer-review editor: @davidslessor




Pingback: PAMPer – The Bottom Line