TRANSFUSE
Age of Red Cells for Transfusion and Outcomes in Critically Ill Adults
Cooper DJ. NEJM 2017; September 27. doi:10.1056/NEJMoa1707572
Clinical Question
- In critically ill patients requiring transfusion, does the transfusion of the freshest available red cells compared to standard care impact on patient mortality?
Background
- When blood is stored there is concern that the storage lesion impacts on patient safety. Red cells become spiculated over time impacting on their transit through capillary beds and oxygen carriage. Hospital donation services need guidance as to safe storage times. There is some variation across centres ranging from 35-42 days. In addition it is not known if certain subgroups of patients would benefit from receiving fresher blood compared with long-term storage of blood.
- There have been several studies investigating whether fresh blood is superior to blood near its used by date. The ABLE trial looked at ICU patients and randomised them to receive blood that was less than 8 days old or older blood. However, there were several limitations to this study, including a relatively small sample size and exclusion of more than 700 potentially eligible patients due to lack of availability of ‘fresh blood.’
- The INFORM trial was a large trial of over 10000 all hospital patients with a subgroup of critically ill patients. Similarly patients were randomized to fresh or old blood. The critically ill subgroup had a lower than expected mortality making it difficult to generalize the results to a sick ICU population.
Design
- Randomised controlled trial 1:1 allocation
- Double blind (concealment of blood collection and expiration dates)
- Computer-generated allocation concealment
- Intention to treat analysis
- ICU patients; prespecified supgroups
- APACHE III with analysis of death above and below the median
- ABO blood group; O vs non-O
- SOFA
- Logistic regression to determine heterogeneity between intervention and control groups
- Further secondary analysis
- analysis of impact of duration of storage independent to group allocation in quartiles and as a continuous variable
- Sample size calculation: 4664 patients required to detect an absolute difference of 4.2% for 90 day day mortality, from a baseline of 28%, with a false positive rate of 10% and a false negative rate of 5%.
Setting
- 59 sites in 5 countries (Australia, New Zealand, Ireland, Finland, Saudi Arabia)
- Dates: November 2012 – December 2016
Population
- Inclusion: patients hospitalized in ICU expected to stay at least 24 hours, who need transfusion (as decided by caring doctors) of at least one RBC unit
- Exclusion
- Age < 18
- Previous RBC transfusion during current admission
- Transplant or haematologic diseases
- Pregnancy
- Cardiac surgery patients (due to low mortality rates)
- Death imminent (< 24 hours)
- Objection by treating physician
- Known patient objection to receive transfusion
- 4994 randomized; 2490 assigned to short-term storage blood, 2504 assigned to long-term storage blood
- Comparing baseline characteristics of control vs. intervention group:
- Baseline characteristics were similar although the age differed by 1 year which was a statistically significant difference (due to the large sample size). The data was therefore analyzed with an adjustment to this age difference.
- Number of red cell units transfused: 4.1 vs. 4.0
Intervention
- Transfusion of the freshest available blood
- Mean storage duration of transfused blood 11.8+-5.3 days
Control
- Transfusion of the oldest available compatible blood (<36 days old, or <42 days in Australia/Saudi Arabia)
- Mean storage duration of transfused blood 22.4 +-7.5
Management common to both groups
- Blood was leukodepleted
- All other management was as per the ICU treating teams
- The mean transfusion threshold was 77.4g/L in the short-term storage group and 77.3g/L in the long-term group.
- After ICU discharge if patients required further transfusion the original allocation assigned would be adhered to.
Outcome
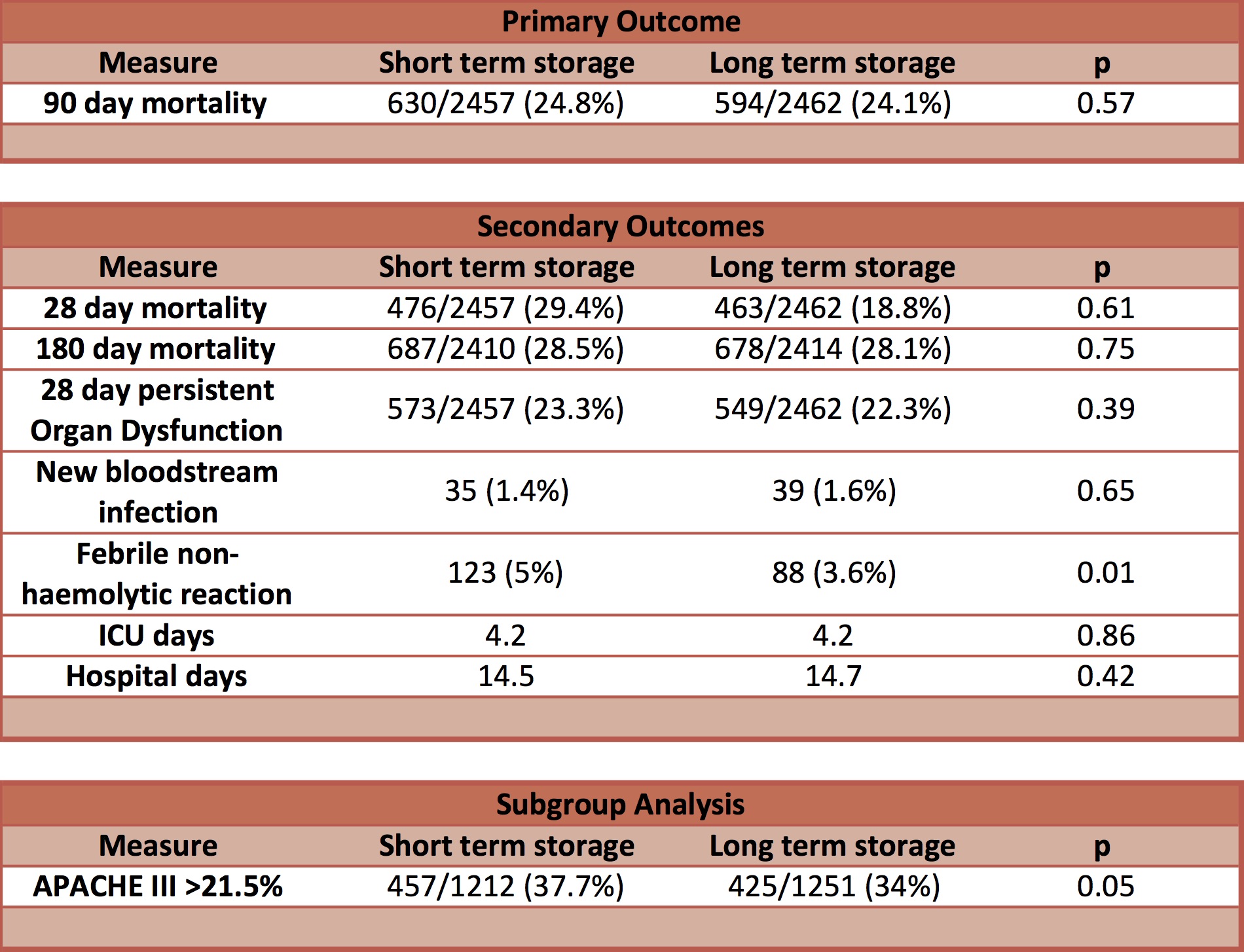
- Primary outcome: Death at 90 days – no significant difference
- Short-term storage 24.8%. Long-term storage 24.1%. unadjusted OR 1.04 (0.91-1.18, p=0.57), adjusted OR 1.05 (0.92-1.21, p=0.46)
- Secondary outcomes: (Short-term vs Long-term blood)
- No significant difference in:
- 28 or 180 day mortality
- Persistent Organ Dysfunction combined with death at day 28
- Days alive and free of mechanical ventilation at Day 90
- Days alive and free of renal replacement therapy at Day 90
- Blood stream infection in ICU
- Length of ICU and hospital stay
- Febrile non-haemolytic tranfusion reactions; occurred more frequently in the short-term storage group (123 events {5%} vs 88 events {3.6%}), absolute risk difference = 1.4% (95% CI, 1.07-1.88, p=0.01)
- Analysis of quartiles depending on age of transfused blood and independent of group allocation: no significant difference
- No significant difference in:
- Sub-group analysis
- Analysis of patients with the higher risk of death (APACHE III > than the median of 21.5%):
- 90 day mortality significantly greater in the short-term storage group
- 37.7% vs. 34%, OR 1.18 (95% CI, 1.00-1.39, p = 0.05)
- 90 day mortality significantly greater in the short-term storage group
- ABO-group transfusion: no significant difference
- SOFA score <7 or >7: no significant difference
- Analysis of patients with the higher risk of death (APACHE III > than the median of 21.5%):
Authors’ Conclusions
- There is no difference in 90 day mortality in patients transfused the freshest available red cells versus standard issue
- Patients who had short-term storage blood had more febrile non-haemolytic transfusion reactions
- Patients who had short-term storage blood with greater than median APACHE III risk of death had increased mortality
Strengths
- Large sample size, meticulous design and execution of the trial ensure these results are the best evidence we have for this clinical question
- Near complete follow-up and analysis adjusted to baseline difference of age
- Only 1% of patients did not receive the blood they were suppose to
- Multi-centred and Multi-country
- The age of the blood in the groups was significantly separated by 10.6 days meaning the trial achieved what it aimed to
Weaknesses
- 1353 eligible patients did not undergo randomisation
- The finding of more febrile non-haemolytic reactions in the fresh blood group should be considered exploratory
- The finding of higher mortality in the sicker patient group is of unknown significance
The Bottom Line
- This important study confirms that there is no benefit gained from using the freshest blood available in the blood bank for transfusing critically ill patients. Conversely it also confirms that there is no harm from using the oldest blood in the blood bank to transfuse critically ill patients
- Although this study will not change my practice it puts the question of “Is Fresh Best” to rest
External Links
- [article] TRANSFUSE: Age of Red Cells for Transfusion and Outcomes in Critically Ill Adults
- [further reading] ABLE
- [further reading] INFORM trial
Metadata
Summary author: Celia Bradford
Summary date: 28th September 2017
Peer-review editor: Segun Olusanya



