BigPAK-2 – preventive care strategy to reduce acute kidney injury after major surgery

A preventive care strategy to reduce moderate or severe acute kidney injury after major surgery (BigpAK-2); a multinational, randomised clinical trial
Zarbock. The Lancet 2025. doi: 10.1016/S0140-6736(25)01717-9
Clinical Question
- In patients at high risk of AKI undergoing major surgery, does a preventative care strategy based on KDIGO guidelines, compared to standard care lead to a reduction in moderate/severe AKI within 72h post-surgery?
Background
- AKI is a common complication of major surgery, associated with increased morbidity and risk of CKD
- Guideline recommended preventative strategies are not routinely implemented
- The KDIGO guidelines recommend implementing specific supportive measures to reduce haemodynamic changes, nephrotoxic exposures and inflammation
- Smaller preliminary studies have suggested implementation of the KDIGO guidelines can reduce incidence of AKI
Design
- Multicentre RCT
- Investigator initiated, open-label
- Adaptive design – Interim analysis of data at pre-specified point for sample size recalculation to ensure power greater than 80%
- Population enrichment with urinary biomarker screening
- Biomarkers TIMP-2 and IGFBP7 used to identify patients at high risk for moderate or severe AKI
- Urinary TIMP-2 x IGFBP7 of < 0.3 (ng/ml)^2/1000 has NPV of 96.3% for moderate/severe AKI
- Important given low rates of post-op AKI in general population (6.7% general surgery and 24.2% in cardiac surgery)
- Randomisation: 1:1 into intervention or control via a web-based system with use of computer-generated, permuted block sequences
- Stratification: According to site
- Blinding: Participants and staff unblinded given nature of intervention but outcome assessors blinded to treatment groups
- Telephone follow up at 30- and 90-days
- Amendments:
- Extension of period of biomarker measurement for 0-4h post-op to 4-18h
- Inclusion of up to 500 cardiac surgical patients
- Primary outcome: Proportion of patients who developed moderate or severe AKI (KDIGO stage 2-3) within 72h of major surgery:
- Serum creatinine > 2.0 times baseline (pre-operative measurement as baseline) and/or urine output <0.5ml/kg/h for >12 hours
- Power Calculation and analysis:
- Based on hypothesised event rates of 20% in the control group and 14% in intervention (based on BigpAK trial)
- Adaptive plan with an interim analysis for sample size recalculation to ensure power of 80%
- Intention-to-treat and per-protocol analyses
- Additional sensitivity analyses for each modular component of KDIGO preventative strategy
Setting
- 34 hospitals across Europe (France, Belgium, Germany, Italy, Netherlands, Spain, UK)
- Nov 2020 -> Jun 2024
Population
- Inclusion:
- Adults
- Major surgery (>2h long, expected ICU/HDU admission post)
- ICU/HDU admission
- IDC and CVC
- At least one risk factor for AKI:
- >= 75 years old
- Ongoing post-op vasopressor and/or mechanical ventilation requirement
- Pre-existing CKD stage 3 (eGFR 30-59)
- Intraoperative radiocontrast agents
- Urinary biomarkers present
- TIMP-2xIGFBP7 conc of >0.3/1000 (ng/ml)^2
- Within 4-18 hours post-surgery
- Exclusion:
- Pre-existing advanced CKD stage 4-5 (eGFR < 30)
- Renal transplant within past 12 months
- RRT within the past 90 days
- Pre-existing anuria, AKI or indications for RRT
- Known glomerulonephritis, interstitial nephritis or vasculitis
- Urinary biomarkers not present
- Participant numbers: 7873 screened -> 1176 included -> 587 intervention, 589 control
- Of those screened but not enrolled (6693) – 4021 no additional AKI risk factor, 1446 had TIMP-2 x IGFBP7 < 0.3, 747 no ICU admission, 575 CKD 4/5, 263 logistical reasons / no biomarker kits available
- Comparing baseline characteristics of intervention vs. control group
- Age: 72 vs 71
- Male: 66.2% vs 66.7%
- Pre-op creatinine (mg/dl): 0.90 vs 0.91
- Medications:
- ACEi: 27.2 vs 25.2%
- ARB: 24.2% vs 24.4%
- CKD stage
- 3a: 15.5% vs 12.4%
- 3b: 7.6% vs 8.1%
- Diabetes:
- Insulin Dependent: 7.1 vs 8.2%
- Non Insulin Dependent: 18.3 vs 15.6%
- ASA score > 2: 78.5% vs 77.7%
- Median APACHE II: 14 vs 15
- Median baseline TIMP-2 x IGFBP7: 0.65 vs 0.66
- Surgery
- Emergency: 10.8% vs 8.8%
- General Surgery: 33.5% vs 35.5%
- Cardiac: 34.0% vs 31.6%
- Vascular: 15.0% vs 15.9%
- Intra-operative management
- Crystalloid Fluid administration (ml): 2243 vs 2366
- Total fluid balance (ml): 1920 vs 1998
- Vasopressors (median cumulative dose to ICU admission)
- Noradrenaline (microg): 1720 vs 1590
- Adrenaline (microg): 491 vs 592
- Vasopressin (IU): 9.8 vs 4.8
- Dobutamine (mg): 27.7 vs 28.2
Intervention
- Preventative KDIGO-recommended care
- Expectation of adherence to all study interventions unless deemed inappropriate by treating clinician
- These consisted of:
- Haemodynamic optimisation for at least 12h post-randomisation
- Passive leg raise manoeuvre 3-hourly to assess fluid-responsiveness
- If positive (CO incr by > 10%) then for 0.5-1L crystalloid bolus
- Targeting MAP > 65 mmHg
- Use of vasopressors to achieve this if necessary
- Passive leg raise manoeuvre 3-hourly to assess fluid-responsiveness
- Advanced haemodynamic monitoring targeting CI > 2.5 ml/min/m^2
- Methods: transpulmonary thermodilution, pulse contour analysis, PAC, TTE/TOE (at discretion of treating clinician)
- Use of dobutamine/epinephrine if necessary
- Withholding nephrotoxic medications
- ACEi and ARBs withheld at least 48h post-op
- Other potential nephrotoxics withheld for 72h if possible
- Tight BSL control (5.5-8.3 mmol/L) – Insulin infusion if required
- Haemodynamic optimisation for at least 12h post-randomisation
Control
- Standard care as per institution
Management common to both groups
- Each site received training in the KDIGO recommended interventions in a site-specific initiation visit
- Pre and intraoperative care standard care as per institution
Interventions Received in both groups
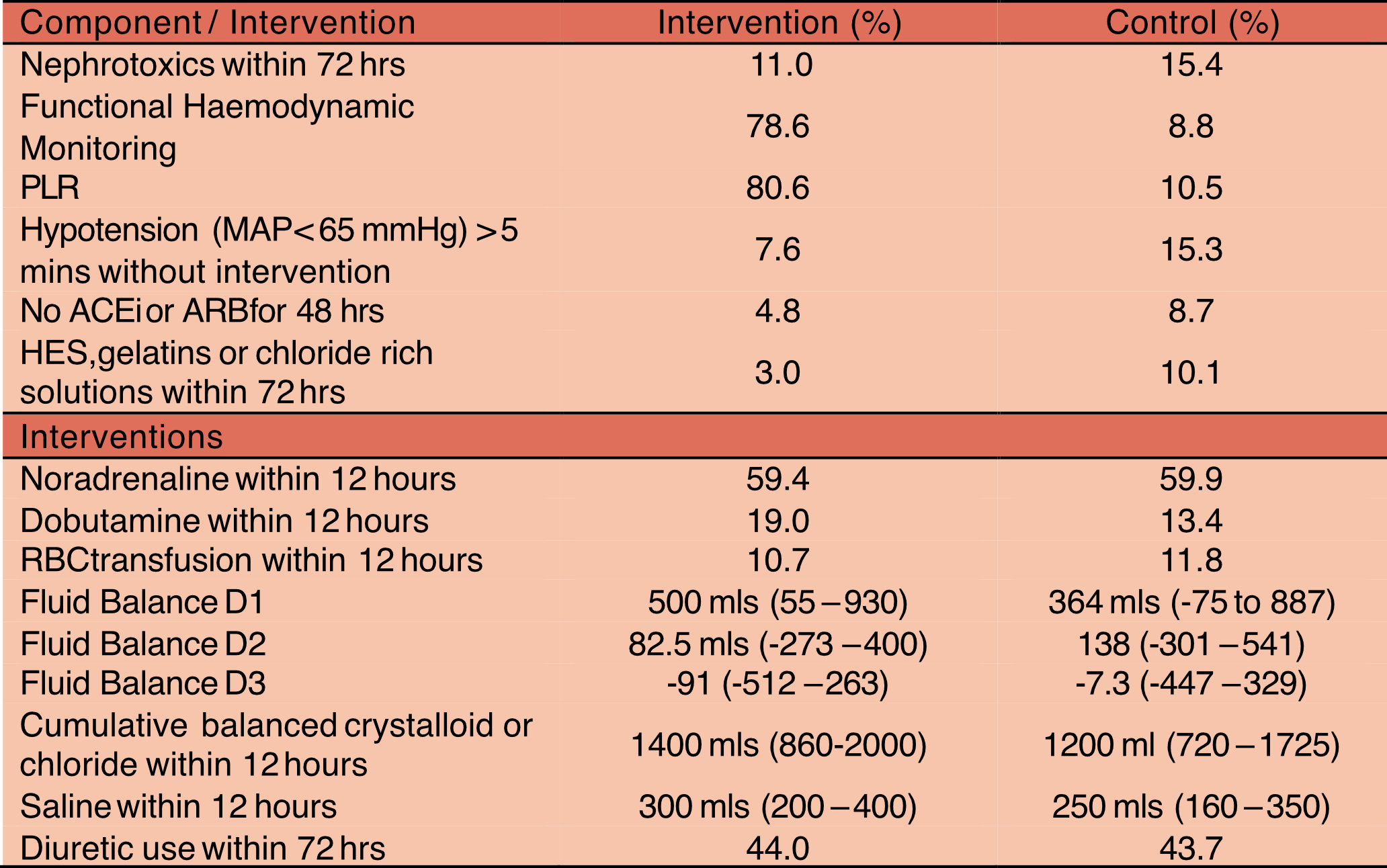
- Taken from Table S6 and S8
Outcome
- Primary outcome:
- Intervention 14.4% (84/587) vs Control 22.3% (131/589)
- OR 0.57 (95% CI 0.40 – 0.79) p=0.0002
- No difference in any subgroups (Sex, CKD 0-2 vs 3), Surgical type, biomarker measurement timing (< 9, > 9 hr)
- Secondary outcomes:
- Less in KDIGO-recommended care group
- Stage 2 AKI (23.5% vs 38.8%, Absolute Difference -15.3%; 95% CI -23.7 to -6.9%)
- Greater in KDIGO-recommended care group
- Stage 1 AKI (60.6% vs 45.4%, Absolute Difference 15.2%; 95% CI 6.1 to 24.2 %)
- Full KDIGO adherence
- Intervention 46.9% vs Control 5.0%, OR 11.58 (7.16 – 18.73)
- No difference:
- AKI (any stage) within 72h
- Intervention 36.5% vs Control 40.8%, OR 0.78 (95% CI 0.6 – 1.01)
- Stage 3 AKI:
- 16.0 vs 15.8%
- Persistent (>48H) moderate or severe AKI
- 39.0% vs 44.5%
- Change in biomarker values 12h post initial measurement
- RRT up to day 90
- 5.1% vs 5.9%
- 90 day mortality
- 7.0% vs 7.0%
- ICU or hospital length of stay
- Days without vasopressor support (to day 3)
- Days without mechanical ventilation (to day 3)
- MAKE 90
- 11.0% vs 10.6%, OR 1.026 (0.692 – 1.522)
- Adverse events
- AKI (any stage) within 72h
- Multivariable analysis
- Association of individual components of KDIGO guidelines with reduction in odds of primary outcome (exploratory only):
- Avoidance of hypotension (MAP < 65): OR 0.19 (95% CI 0.07 – 0.53), (p=0.001)
- Discontinuation of ACEi/ARBs: OR 0.36 (0.19 – 0.70), (p=0.002)
- All other components had 95% CI crossing 1
- Association of individual components of KDIGO guidelines with reduction in odds of primary outcome (exploratory only):
Authors’ Conclusions
- Compared with usual care, in major surgery patients at high risk for AKI, a KDIGO-recommended preventive care strategy significantly decreased the occurrence of moderate or severe AKI within 72 h of surgery
Strengths
- Multicentre RCT across multiple countries
- Study groups well balanced with similar baseline characteristics
- Adaptive design ensured adequate power
- Bias was minimised with computer-generated randomisation and blinding of primary outcome assessors
- Inclusion of multiple surgical specialties increases generalisability
- Clinically relevant primary endpoint
- Primary endpoint demonstrated on ITT, PP and AT analyses
- Minimal loss to follow up with 99.66% of participants included in primary endpoint
- All pre-specified primary and secondary endpoints reported
Weaknesses
- Unblinded participants and clinicians, however, primary endpoint is objective
- Poor protocol adherence in intervention group and some crossover from control group
- < 50% in intervention group received full preventative strategy – increased to 62% without hyperglycaemia management component
- Around 10% in control group had functional haemodynamic monitoring and regular PLR
- Intervention was a cluster of cares with education delivered to staff at each site prior to study, however, randomisation was performed at patient level
- This may account for some unmeasured crossover between groups if staff are implementing both strategies for different patients; however one would expect this to bias towards the null
- The control arm consisted of standard care, with potential heterogeneity of practice across multiple centres and countries
- Cardiac surgical patients are at significantly higher risk of post-op AKI when compared to general surgical patients (24.2% vs 6.7%)
- Exclusion of biomarker negative patients and patients with Stage 4-5 CKD limits generalisability and effect size likely much smaller in general population that a clinician would see on a day to day basis
- Unclear which component(s) of the bundle had greatest effect
- On exploratory analysis avoidance of ARB/ACEi and hypotension seem key
- Despite monitoring it appears that the actual differences in haemodynamic interventions are minimal (increased use of dobutamine and a median difference of around +200mls fluid within 12 hours)
- Potential that it is an ancillary effect of regular clinician review and examination of patients that has occurred in the implementation of this bundle
- Whilst rates of Stage 2/3 AKI reduced it, no change in any AKI (largely as increased rates of Stage 1 AKI in intervention group). This is important if Stage 1 AKI has clinically important longer term effects
The Bottom Line
- This RCT demonstrated that adherence to the KDIGO guidelines is associated with a reduction in the rate of moderate to severe AKI in the first 72h following major surgery. However, it did not demonstrate a difference when compared to standard care with longer term, patient orientated outcomes
- It appears AKI risk can be reduced from high-quality supportive care with frequent patient assessment
External Links
- article A preventive care strategy to reduce moderate or severe acute kidney injury after major surgery (BigpAK-2); a multinational, randomised clinical trial
- editorial Bundled interventions to prevent acute kidney injury: finding the value in supportive care
- FOAMEd NephJC
Metadata
Summary author: Patrick MacNamara
Summary date: 12th March 2026
Peer-review editor: George Walker
Picture by: Pexels