EUSOS
EUSOS: Mortality after surgery in Europe
Pearse et al. Lancet 2012; 380:1059-65. doi:10.1016/S0140-6736(12)61148-9
Clinical Question
- What is the clinical outcome of patients after major non-cardiac surgery in Europe?
Design
- Prospective cohort design
- Participating hospitals were a voluntary convenience sample
Setting
- 498 hospitals across 28 European countries (56% university affiliated)
- 7 days in April 2011
Population
- Inclusion: all adults (over 16 years) undergoing elective or non-elective inpatient surgery
- Exclusion: day-case surgery; cardiac surgery; neurosurgery; radiological procedures; obstetric procedures
- 46,985 patients screened, 46,539 included in analysis (1% removed: duplicate, inconsistent or missing data)
Outcome
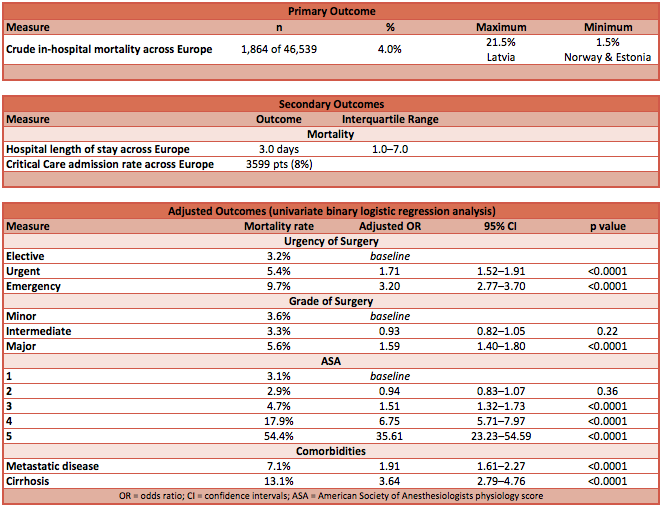
- Primary outcome:
- Crude in-hospital mortality across Europe was 4.0%
- Compared to UK as baseline, four countries had statistically significantly different and unexplained higher rates of mortality:
- Poland — adjusted OR 6.92 (95% CI 2.37–20.27); p-value 0.0004
- Romania — adjusted OR 3.19 (95% CI 1.61–6.29); p-value 0.001
- Latvia — adjusted OR 4.98 (95% CI 1.22–20.29); p-value 0.025
- Ireland — adjusted OR 2.61 (95% CI 1.30–5.27); p-value 0.007
- Compared to UK, no country had statistically significantly better rates of mortality, although UK was 10th in rank order
- Secondary outcome:
- Hospital stay 3.0 days [IQR 1.0 – 7.0]
- Overall admission rate to ICU was 8%
- Of which it was unplanned for 29%
- Mortality in this unplanned Critical Care cohort was 23% compared to 10% in the planned Critical Care cohort
- Median critical care stay was 1.2 days [IQR 0.9 – 3.6]
- Of which it was unplanned for 29%
- Of all the deaths, 73% of these patients never attended Critical Care
- Of the deaths in the cohort that did attend Critical Care, 43% occurred after discharge from Critical Care
- Independent variables associated with increased mortality:
- Country
- Urgency
- Grade of surgery
- Surgical procedure category
- American Society of Anesthesiologists physiology score (ASAps)
- Metastatic disease
- Cirrhosis
 |
| Summary of EuSOS Data |
 |
| Choropleth map graphically illustrating primary and secondary outcomes by country |
Authors’ Conclusions
- The findings demonstrate that 4% of patients undergoing inpatient surgery die before discharge from hospital, which was higher than expected and varies substantially across Europe.
- There is a systematic failure in the allocation of Critical Care resources
Strengths
- First, large, multi-centre, multi-national cohort survey
- Only small number of cases excluded due to missing data
- Outcomes are comparable to smaller national surveys
Weaknesses
- May not represent true outcomes across Europe, as only based on self-selecting hospitals
- Some countries reported only small numbers of patients, so the described practice and outcomes may not represent national practice
- True number of eligible patients not known, as some may not have been reported
- Risk-adjustments made for confounding variables were based upon subjective scores such as ASA and surgical urgency category
- As a cross-sectional survey, associations and causations are difficult / impossible to conclude with certainty
The Bottom Line
- This landmark cross-sectional survey demonstrated that in-hospital mortality is higher than perceived, that unexpected variation exists across Europe, and Critical Care is poorly resourced given that many deaths happened in those that did not attend Critical Care or attended as an unplanned admission
External Links
- [pubmed] Mortality after surgery in Europe: a 7 day cohort study
- [article] Mortality after surgery in Europe: a 7 day cohort study
Metadata
Summary author: @DuncanChambler
Summary date: 19 June 2015
Peer-review editor: @DavidSlessor



