ELAIN FOLLOW-UP
Long-Term Clinical Outcomes after Early Initiation of RRT in Critically Ill Patients with AKI
Meersch M. J Am Soc Nephrol 29:Dec 2017 doi:10.1681ASN.2017060694
Clinical Question
- In adult critically ill patients, with acute kidney injury, does early initiation of renal replacement therapy (RRT) compared to delayed initiation of RRT reduce the composite endpoint of MAKE365; consisting of death, RRT and persistent renal dysfunction at one year?
Background
- The optimal timing for initiation of RRT in patients with AKI is unknown.
- This trial looked at the 1 year outcomes of patients enrolled in the ELAIN trial.
- The ELAIN trial examined 90 day mortality of patients who were randomised to receive early RRT (within 8 hours of reaching KDIGO Stage 2) or delayed RRT (within 12 hours of reaching KDIGO Stage 3) or if they had an absolute indication for initiation of RRT.
- The ELAIN trial showed improved 90 day mortality (early vs delayed RRT) of 39.3% vs 54.7%, shorter duration of RRT (9 days vs 25 days), reduced ventilator time (125 hrs vs 181 hrs) and reduced hospital LOS (51 days vs 82 days).
- This trial aimed to determine if the beneficial intervention of early RRT was sustained at the 1 year mark.
- It is known that the mortality and morbidity after receiving RRT in ICU is high. For example, the POST-RENAL (prolonged outcomes after kidney injury requiring dialysis in the ICU) study suggested that at 4 years more than 60% of patients who had received dialysis in ICU were now dead and more than 5% of those who survived required ongoing dialysis.
- The STAART-AKI trial is currently recruiting. This trial is larger than ELAIN as it is aiming to recruit 2866 patients (vs 231 for ELAIN). It is comparing early RRT vs standard care. This study includes ICU adults with renal dysfunction who have i) A doubling of baseline creatinine OR ii) Cr >354umol/L OR iii) UO<0.5ml/kg/hr for 12 hours. The primary outcome is 90 day mortality and independence from RRT.
Design
- The ELAIN trial was randomised, unblinded, single-centre with complete follow-up. Patients were allocated 1:1 for early vs late dialysis stratified by hemodynamic SOFA score.
- For patients discharged alive at 90 days, telephone calls were made to the general practitioner, subject or family member to determine if the patient was alive, if they were requiring RRT or if they had persistent renal dysfunction (defined as 25% decline in eGFR compared with baseline calculated with the Cockroft-Gault formula)
- In addition the relation of inflammatory biomarkers (collected at ICU Day 0 and Day 3) to the MAKE365 outcome was analysed.
- 1 year outcomes were available for all but one patient
Setting
- University Hospital Munster, Gemany
- ELAIN recruited from August 2013 to December 2015
Population
- Inclusion: must have met all five of the following
1. Acute kidney Injury- KDIGO stage 2:
- 2-fold increase in serum creatinine from baseline, or urine output <0.5ml/kg/hr for ≥12 hrs despite optimal resuscitation (PCWP>12, CI >2.6, MAP>65, IAP<15)
2. NGAL >150ng/dl
3. At least one of the following conditions:
- Severe sepsis
- Catecholamines (noradrenaline/adrenaline) >0.1mcg/kg/min
- Refractory fluid overload (worsening pulmonary oedeoma, P/F <300mgHg, fluid balance >10% body weight)
- Development or progression of non-renal organ dysfunction (SOFA organ system score ≥2)
4. Age between 18 and 90
5. Intention to provide full intensive care treatment for at least 3 days
- Exclusion:
-
- Patients with pre-existing kidney disease not requiring RRT
- Patients who had received any previous RRT
- AKI caused by permanent occlusion/surgical lesions of the renal artery
- Obstructive AKI
- HUS/TTP
- Prior Kidney transplant
- Hepatorenal syndrome
- AIDS with CD4 count <0/05 X109/L
- 604 screened, 231 randomised and included in primary analysis
Intervention
- Initiation of RRT within 8 hours of confirmation of KDIGO stage 2
- Delivered to 100% of patients assigned to early group
- Median time to RRT was 6.0 hours (IQR 4.0-7.0)
Control
- Inititation of RRT within 12 hours of either
- KDIGO stage 3 criteria
- Creatinine rise >3 fold increase from baseline, or oliguria <0.3ml/kg/hr for ≡24 hours or serum creatinine > 4mg/dl (353.6 umol/l) with an acute increase of at least 0.5mg/dl (44.2umol/l) within 24 hours
- Absolute indication for RRT
- Urea >100mg/dl
- Potassium >6mmol/l and or ECG abnormalities
- Magnesium >4mmol/l and/or anuria/absence of deep tendon reflexes
- Blood pH <7.15
- Urine production <200ml/12hr or anuria
- Organ oedema in the presence of AKI resistant to diuretic treatment (defined as one trial of furosemide)
- KDIGO stage 3 criteria
- Delivered to 91% of patients assigned to delayed group
- Median time to RRT was 25.5 hours (IQR 18.8-40.3)
Common to both groups
- RRT standardized
- CVVHDF
- prediction 100%
- dialysate:replacement fluid ratio 1:1
- effluent flow 30ml/kg
- minimum 110ml/min blood flow
- citrate anticoagulation
- RRT discontinued if renal recovery evident by both:
- > 400ml urine in 24hr without diuretics or > 2100ml in 24hr with diuretics
- creatine clearance > 20ml/min occurred
- RRT could be changed to SLED after 7 days if no recovery
- Additional treatments (ventilation, nutrition) standardized
- Follow up up to 1 year post randomization
Outcome
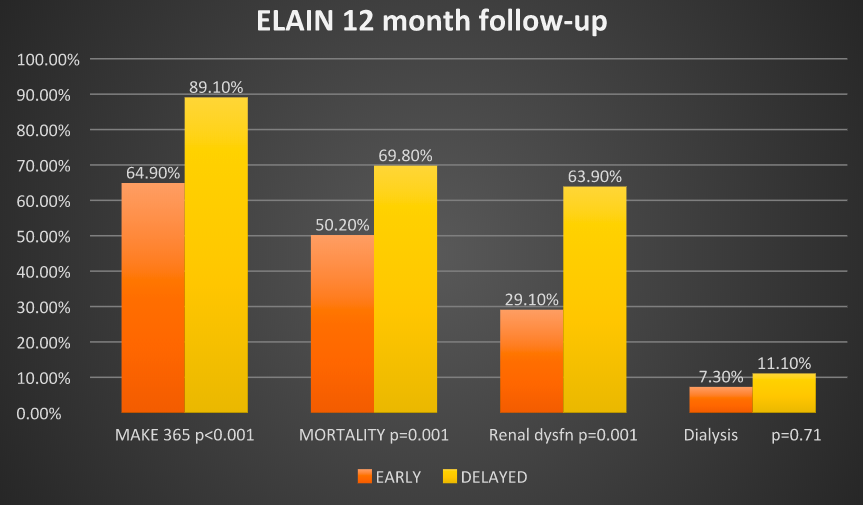
- Primary outcome: MAKE365 score at 1 year (a composite of persistent renal dysfunction, dialysis dependence and mortality) [Early vs Delayed]
- 64.9% vs 89.1% p<0.001 (OR 0.23 [0.11-0.24])
- Fragility index: 17
- Number needed to treat: 5
- Secondary outcomes:
- 1 year all cause mortality [Early vs Delayed]
- 50.2% vs 69.8% p<0.01 (OR 0.62 [0.44-0.87])
- Increase in mortality between day 90 and 1 year
- Early group: 39.3% to 50.2%
- Delayed group: 54.7% to 69.8%
- Failure to recover renal function p=0.001
- Early group: 29.1% (16 of 55)
- Delayed group: 63.9% (23 of 36)
- Need for RRT at 1 year p=0.71
- Early group: 7.3% (4 of 55)
- Delayed group: 11.1% (4/36)
- 1 year all cause mortality [Early vs Delayed]
- Exploratory biomarker analysis: 159 of 204 patients had biomarkers measured on day 0 and 3 after randomisation (Pro-inflammatory: MIF, IL-6, IL-8 and IL-18 and Anti-inflammatory: IL-10 and TNFR-1 and TNFR-2)
- Elevated TNFR-1 and TNFR-2 at Day 3 were higher in those who met the MAKE365 end point. Elevations in the other markers were not associated with MAKE365 positive group.
Authors’ Conclusions
- In this cohort of critically ill patients with AKI, after 1 year, early initiation of RRT compared with delayed RRT continues to have a benefit on a composite outcome of mortality, dialysis dependence and renal function.
Strengths
- Only one patient lost to follow-up at 1 year
- An important question: is RRT in the ICU setting merely supportive or can it influence patient centred outcomes and does the timing of initiation influence this?
- Intention to treat analysis; those in the delayed group who did not receive dialysis (11 patients; 6 did not progress to Stage 3, 4 reached Stage 3 but did not get dialysis, 1 did not get dialysis as no machine available) were analysed in the group they were randomised to
Weaknesses
- Single centred
- Unblinded
- Mainly post-surgical cohort with high proportion of post-cardiac surgery cases, making the generalisability difficult
- The study is small and likely underpowered. The fragility index of the original ELAIN study was only 3.
- The primary outcome was not included in the trial protocol. The only 1 year outcome pre-specified in the trial protocol was all cause mortality.
The Bottom Line
- The results suggest that early RRT, in predominantly post-operative patients with Stage 2 AKI, positively influences mortality and other patient centred outcomes at 1 year.
- Renal replacement therapy does carry cost and some risk to patients, so although this encourages me to use RRT early, I await the results of the STAART-AKI trial, which will be invaluable in answering the important question of the optimal timing of RRT in critically ill patients.
External Links
- [article] Long-Term Clinical Outcomes after Early Initiation of RRT in Critically Ill Patients with AKI
- [further reading] Effect of Early vs Delayed Initiation of Renal Replacement Therapy on Mortality in Critically Ill Patients With Acute Kidney Injury: The ELAIN RCT
- [further reading] Early vs Late RRT: editorial TBL
Metadata
Summary author: @Celia Bradford
Summary date: 9/2/18
Peer-review editors: Segun Olusanya @iceman_ex and @davidslessor



